More than 600 Corby trial volunteers give new Novavax vaccine a massive shot in the arm
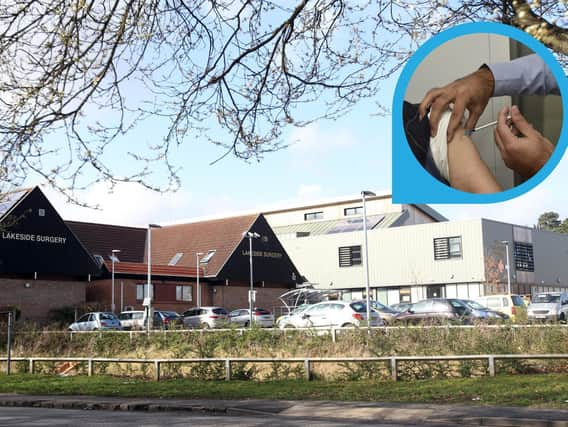

Volunteers who came forward to trial a new Covid vaccine in Corby have been praised for their 'fantastic response' in stepping up to test the life-saving jab.
GPs at Lakeside Healthcare in Cottingham Road recruited more than 600 people to trial the Novavax Covid-19 vaccine, with half being given a placebo and half the real vaccine - in two doses.
Advertisement
Hide AdAdvertisement
Hide AdResults show that the ground-breaking Covid vaccine to be nearly 90 per cent effective trial amongst the over 15,000 participants recruited from 35 research UK sites.
Dr Amardeep Heer, GP Partner and Director of Research and Innovation at Lakeside Healthcare said: “This is a great achievement and is directly due to the fantastic response we received from patient volunteers who came forward to take part in this trial at Lakeside Corby.
"Together, the hard work of the Research Delivery team and the enthusiasm of the trial volunteers have allowed us to reach this very important milestone.
“I would like to thank all of the patient volunteers who contacted us and gave up their valuable time to take part together with all of the Lakeside and Clinical Research Network Staff who have worked so tirelessly on this trial.”
Advertisement
Hide AdAdvertisement
Hide AdThe NIHR-supported Novavax Covid-19 vaccine is 89.3 per cent effective at preventing Covid-19, shown in early analysis of its Phase III study data, including effectiveness against the new mutant variants.
The Novavax study is the largest ever double blind, placebo-controlled trial to be undertaken in the UK. Half were given two intramuscular injections of the vaccine 21 days apart and half a saline solution.
Gordon Huxtable, an IT manger from Corby volunteered for the trial at the surgery where his sister is a nurse so that he could help beat the virus.
He said: "I'm really proud to be part of it. The fact that hundreds of people in Corby took part and have played a part in this vaccine show that the people in the town have hearts of gold.
Advertisement
Hide AdAdvertisement
Hide Ad"It was a little bit nerve-wracking at the start but I talked it through with my family and did some research, assessing the risks.
"They are just a really small company that has come up with the answer - no complicated deep freeze or anything like that and it stops transmission of the virus - it's a game changer."
Interim efficacy and safety data will now be submitted to all regulators across the world - including the Medicines and Healthcare products Regulatory Agency (MHRA) in the UK - for independent scrutiny and product approval.
A significant proportion of participants taking part in the study were recruited through the NHS COVID-19 Vaccine Research Registry - an online registry of over 390,000 people who have signed-up to be contacted about taking part in COVID-19 vaccine studies.
Advertisement
Hide AdAdvertisement
Hide AdMore than 25 per cent of enrollees in the trial were over the age of 65, while a large proportion of volunteers had underlying medical conditions generally representative of the population.
Professor Paul Heath, Novavax Phase 3 trial Chief Investigator and Professor of Paediatric Infectious Diseases at St George’s University Hospitals NHS Foundation Trust said: “These are enormously exciting findings and show that this is a highly effective and safe COVID-19 vaccine – importantly it also shows that this is a vaccine that is effective against the UK variant that has spread so quickly. This wonderful news is a tribute to the over 15,000 volunteers in our trial, to the dedication of the UK investigators and to the huge support of the NIHR.”
Clive Dix, Chair, UK Vaccine Taskforce, said: “These are spectacular results, and we are very pleased to have helped Novavax with the development of this vaccine. The efficacy shown against the emerging variants is also extremely encouraging. This is an incredible achievement that will ensure we can protect individuals in the UK and the rest of the world from this virus.”
Mr Huxtable, 51, added: "I've no regrets about the trial. It's great that so many people stepped up to the plate.
Advertisement
Hide AdAdvertisement
Hide Ad"I'm not sure if I had the vaccine or not. I didn't feel anything but I'm going to volunteer for the next phase, a cross-over project, when I believe they will give you the opposite of what you first had. So one way or the other I will get the jab."
For more information about volunteering for future vaccine studies go to www.nhs.uk/researchcontact.The UK government has already secured 60 million doses of the Novavax NVX-CoV2373 vaccine.
Provided it meets standards on safety, effectiveness and quality following publication of results, the vaccine will be manufactured using FUJIFILM Diosynth Biotechnologies’s facilities in Billingham, Stockton-on-Tees.